2020 New Style Candida Albicans Agar - Trichomonas vaginalis – Liming Bio
2020 New Style Candida Albicans Agar - Trichomonas vaginalis – Liming Bio Detail:

INTENDED USE
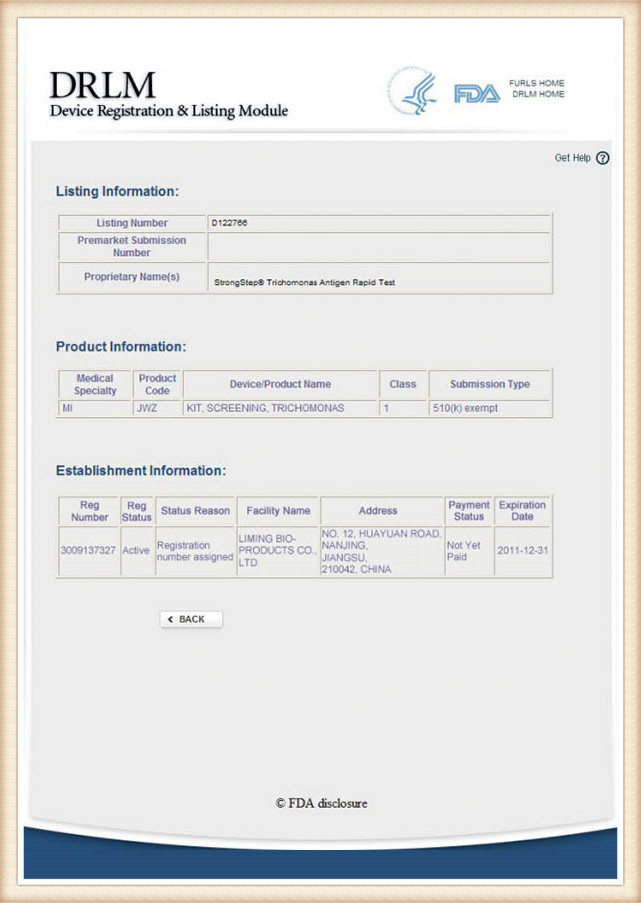
StrongStep® Trichomonas Vaginalis Antigen Rapid Test is intended for the qualitative detection of Trichomonas vaginalis (*Trichomonasw) antigens from vaginal swabs. This kit is intended to be used as an aid in the diagnosis of Trichomonas infection.
INTRODUCTION
Trichomonas infection is responsible for the most common, non-viral sexually transmitted disease (vaginitis or trichomoniasis) worldwide. Trichomoniasis is a significant cause of morbidity among all infected patients. Effective diagnosis and treatment of Trichomonas infections have been shown to eliminate symptoms. Conventional identification procedures for Trichomonas from vaginal swabs or vaginal washes involve the isolation and subsequent identification of viable pathogens by wet mount microscopy or by culture, a process that will cost 24-120 hours. Wet mount microscopy has a reported sensitivity of 58% versus culture. The StrongStep9^ Trichomonas vaginalis Antigen Rapid Test is an immunochromatographic assay that detects pathogen antigens directly from vaginal swabs. Results are rapid, occurring within approximately 15 minutes.
PRINCIPLE
Sfrong5fep®Trichomonas vaginalis Antigen Rapid Test uses dyed latex immunochromatographic, capillary flow technology. The test procedure requires the solubilization of Trichomonas proteins from a vaginal swab by mixing the swab in Sample Buffer. Then the mixed sample buffer is added to the test cassette sample well and the mixture migrates along the membrane surface. If Trichomonas is present in the sample, it will form a complex with the primary anti-Trichomonas antibody conjugated to dyde latex particles (red). The complex will then be bound by a second anti-Trichomonas antibody coated on the nitrocellulose membrane. The appearance of a visible test line along with the control line will indicate a positive result.



Trichomonas 1
Trichomonas 2
Instructions
MSDS
Product detail pictures:


Related Product Guide:
The organization keeps for the procedure concept "scientific administration, superior quality and effectiveness primacy, shopper supreme for 2020 New Style Candida Albicans Agar - Trichomonas vaginalis – Liming Bio , The product will supply to all over the world, such as: Italy, Salt Lake City, Algeria, We will not only continuously introduce technical guidance of experts from both home and abroad, but also develop the new and advanced products constantly to satisfactorily meet the needs of our clients all over the world.
The goods are very perfect and the company sales manager is warmful, we will come to this company to purchase next time.