China Cheap price Sars-Cov-2 Detect - SARS-CoV-2 Antigen Rapid Test for Saliva – Liming Bio
China Cheap price Sars-Cov-2 Detect - SARS-CoV-2 Antigen Rapid Test for Saliva – Liming Bio Detail:
INTENDED USE
The StrongStep® SARS-CoV-2 Antigen Rapid Test is a rapid immunochromatographic assay for the detection of SARS-CoV-2 virus Nucleocapsid Protein antigen in human Saliva collected from individuals who are suspected of COVID-19 by their healthcare provider within the first five days of the onset of symptoms. The assay is used as an aid in the diagnosis of COVID-19.
INTRODUCTION
The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
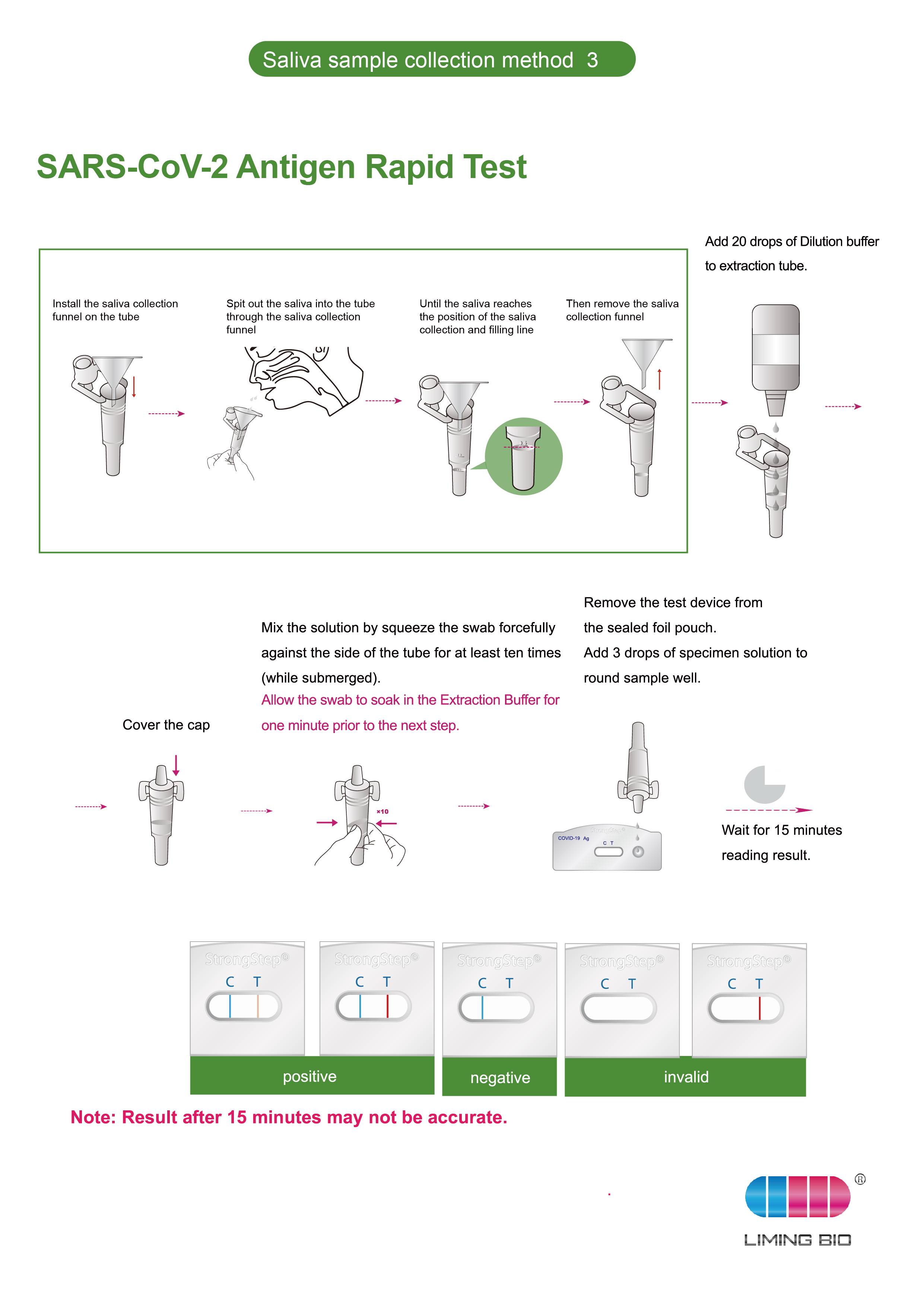
Product detail pictures:
1人份抗原卡实物图唾液版1_00_副本.png)
1人份抗原卡实物图唾液版1_00_副本.jpg)
Related Product Guide:
We depend on sturdy technical force and continually create sophisticated technologies to meet the demand of China Cheap price Sars-Cov-2 Detect - SARS-CoV-2 Antigen Rapid Test for Saliva – Liming Bio , The product will supply to all over the world, such as: Leicester, Korea, Norwegian, As the world economic integration bringing challenges and opportunities to the xxx industry, our company , by carrying on our teamwork, quality first, innovation and mutual benefit, are confident enough to supply our clients sincerely with qualified merchandise, competitive price and great service, and to build a brighter future under the spirit of higher, faster, stronger with our friends together by carrying on our discipline.
This company can be well to meet our needs on product quantity and delivery time, so we always choose them when we have procurement requirements.