Fast delivery Chlamydia Trachomatis Antigen Test - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio
Fast delivery Chlamydia Trachomatis Antigen Test - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio Detail:

The StrongStep® Neisseria gonorrhoeae Antigen rapid test is a rapid lateral-flow immunoassay for the qualitative presumptive detection of Neisseria gonorrhoeae antigen in male urethral and female cervical swab.
Benefits
Accurate
High sensitivity(97.5%) and high specificity(97.4%) according to the results of 1086 cases of clinical trials.
Rapid
Only 15 minutes required.
User-friendly
One-step procedure to detect the antigen directly.
Equipment-free
The source-limiting hospitals or clinical setting can perform this test.
Easy-to-read
Easily-interpreted by all healthcare personnel.
Storage conditions
Room temperature (2℃-30℃), or even higher(stable for 1 year at 37℃).
Specifications
Sensitivity 97.5%
Specificity 97.4%
CE marked
Kit Size=20 kits
File: Manuals/MSDS



Brochure1
Brochure 2
Instructions
MSDS
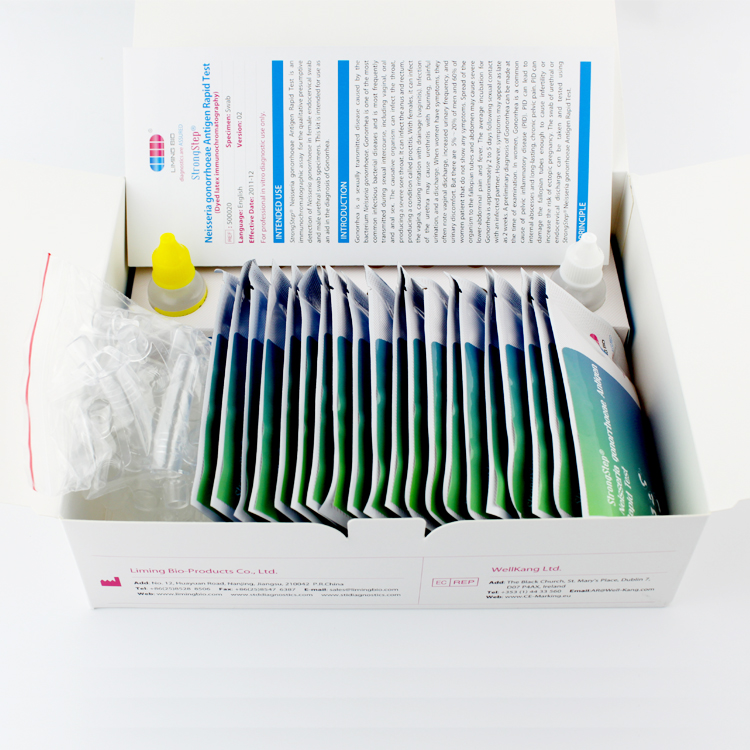
Product detail pictures:




Related Product Guide:
We insist on offering high-quality production with great enterprise concept, honest product sales and also the finest and fast service. it will bring you not only the superior quality solution and huge profit, but the most significant should be to occupy the endless market for Fast delivery Chlamydia Trachomatis Antigen Test - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio , The product will supply to all over the world, such as: British, Jersey, Bangalore, Our company offers the full range from pre-sales to after-sales service, from product development to audit the use of maintenance, based on strong technical strength, superior product performance, reasonable prices and perfect service, we will continue to develop, to provide the high-quality products and services, and promote lasting cooperation with our customers, common development and create a better future.
We have been looking for a professional and responsible supplier, and now we find it.