Wholesale Coronavirus Test Kit Wholeblood - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio
Wholesale Coronavirus Test Kit Wholeblood - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio Detail:

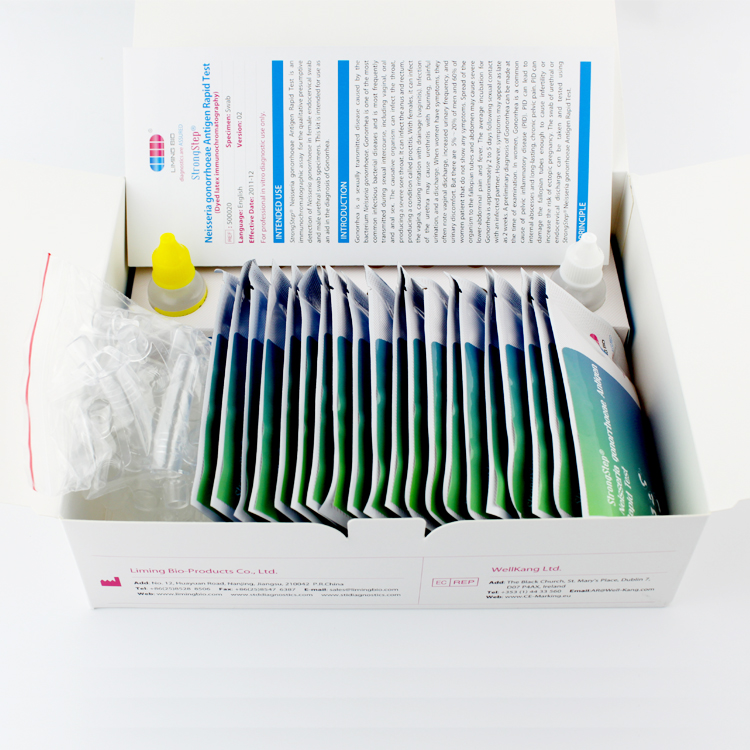
The StrongStep® Neisseria gonorrhoeae Antigen rapid test is a rapid lateral-flow immunoassay for the qualitative presumptive detection of Neisseria gonorrhoeae antigen in male urethral and female cervical swab.
Benefits
Accurate
High sensitivity(97.5%) and high specificity(97.4%) according to the results of 1086 cases of clinical trials.
Rapid
Only 15 minutes required.
User-friendly
One-step procedure to detect the antigen directly.
Equipment-free
The source-limiting hospitals or clinical setting can perform this test.
Easy-to-read
Easily-interpreted by all healthcare personnel.
Storage conditions
Room temperature (2℃-30℃), or even higher(stable for 1 year at 37℃).
Specifications
Sensitivity 97.5%
Specificity 97.4%
CE marked
Kit Size=20 kits
File: Manuals/MSDS



Brochure1
Brochure 2
Instructions
MSDS
Product detail pictures:




Related Product Guide:
We usually perform being a tangible workforce making sure that we will give you the most beneficial excellent plus the finest selling price for Wholesale Coronavirus Test Kit Wholeblood - Neisseria Gonorrhoeae Antigen Rapid Test – Liming Bio , The product will supply to all over the world, such as: Venezuela, Ottawa, Italy, We have been perfectly devoted to the design, R&D, manufacture, sale and service of hair products during 10 years of development. We have introduced and are making full use of internationally advanced technology and equipment, with advantages of skilled workers. "Dedicated to providing reliable customer service" is our aim. We are sincerely looking forward to establishing business relationships with friends from at home and abroad.
With a positive attitude of "regard the market, regard the custom, regard the science", the company works actively to do research and development. Hope we have a future business relationships and achieving mutual success.